Terms and Agreement
Please agree to the terms to continue using the chat feature.


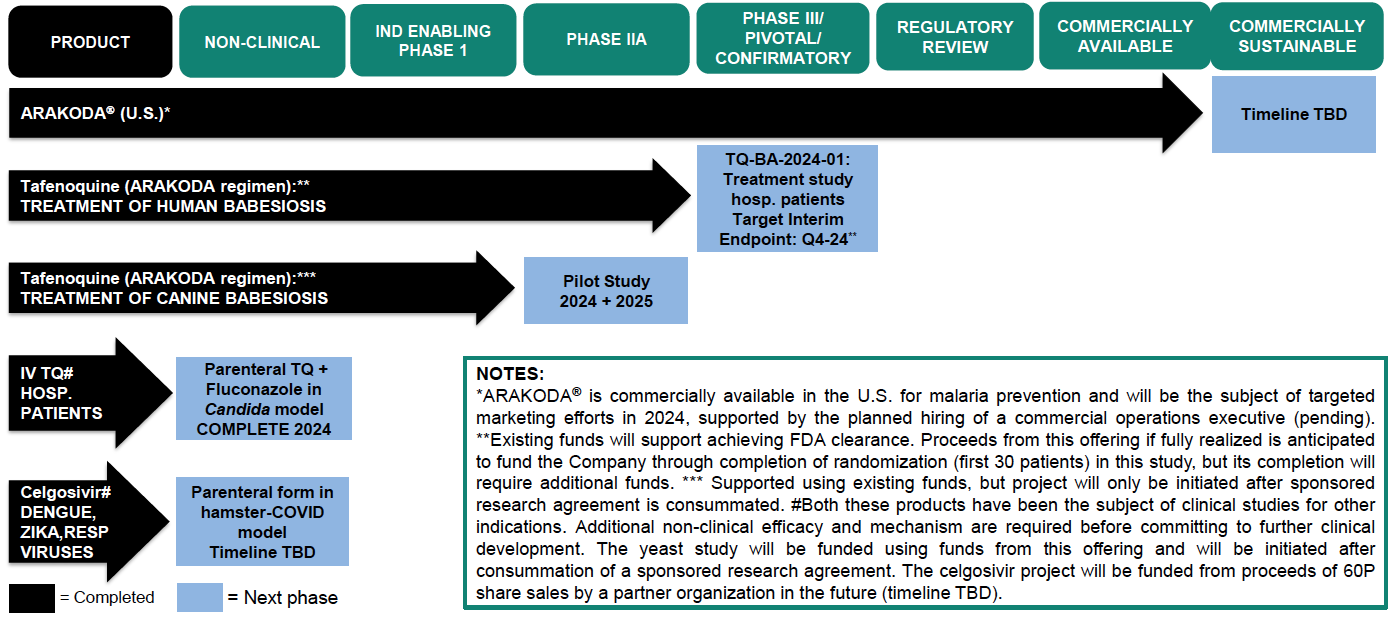
60 Degrees Pharmaceuticals (60P) is a growth-oriented biotechnology company with a goal of using cutting-edge biological science and applied research to further develop and commercialize new therapies for the prevention and treatment of infectious diseases. 60P has successfully achieved regulatory approval of ARAKODA® (tafenoquine), a malaria preventative treatment that has been on the market since late 2019. Currently, 60P’s pipeline includes development programs for Tafenoquine and Celgosivir targeting fungal, tick-borne, and other viral diseases.
60P is addressing the unmet medical need associated with infectious diseases through the development and commercialization of new small molecule therapeutics. By focusing on synthetic drugs (made by chemists in labs, excluding biologics) with good safety profiles based on prior clinical studies, 60P believes it has a cost-effective path to new indications that capitalizes on existing research to reduce costs and risk. 60P is expanding its commercialization efforts related to ARAKODA (tafenoquine), an antimalarial indicated for prophylaxis of malaria in patients 18 years and older and approved by the FDA in 2018. In Q2 2023, sales of ARAKODA increased by 150% relative to the same period in 2022, at an accelerating growth rate.
60P is implementing clinical research programs to evaluate the utility of the ARAKODA regimen of tafenoquine for non-malaria disease indications, with an upcoming planned pivotal study of tafenoquine in hospitalized babesiosis patients. 60P anticipates initiating patient enrollment for this trial in mid-2024. According to Company estimates, 47,000 cases of babesiosis (infections caused by red blood cell parasites similar to malaria that are transmitted by deer tick bites) occur in the United States each year, and the incidence rate is increasing. Estimates are that 10% of Lyme disease patients are co-infected with babesiosis. 60P is also testing the viability of another product (Celgosivir) to determine whether to advance it into further clinical development and may seek to develop and license other molecules in the future.
(Pivotal Babesiosis Study)
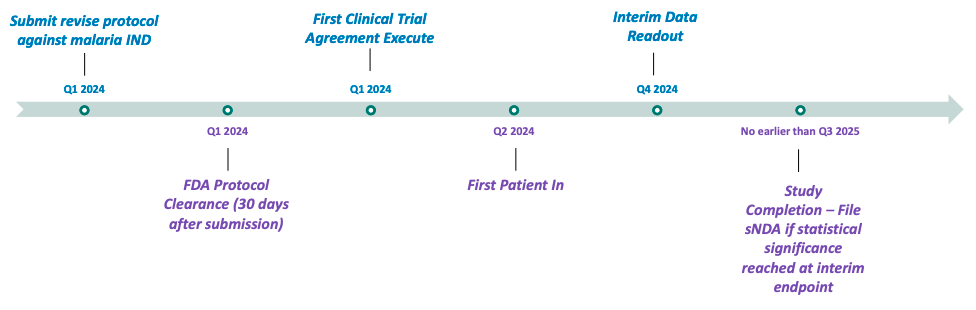
Other Anticipated Milestones:
Red Chat is here to summarize the SEC filing data on SXTP
Please agree to the terms to continue using the chat feature.